On January 25, 2023, Science Daily released an article about new research discovered by Osaka Metropolitan University regarding the Synthesis of fumaric acid by a new method of artificial photosynthesis, using sunlight to make biodegradable plastic.
Global warming has caused a growing issue in our environment due to greenhouse gasses such as CO2. This research states that by using artificial photosynthesis CO2 can be reduced, hence limit global warming. This discovery shows that fumaric acid can be synthesized from CO2 and biomass-derived compounds using renewable solar energy.
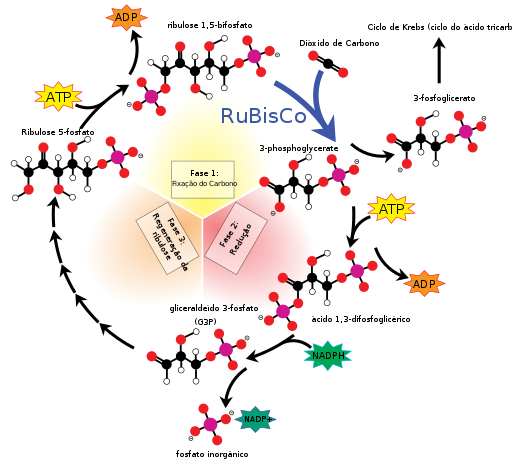
As we have learned in Biology class, photosynthesis is an anabolic reaction because it builds up glucose, a bigger molecule, from water and carbon dioxide. Although –overall– photosynthesis is an anabolic reaction, catabolic reactions occur throughout photosynthesis because the large molecules, CO2 and H2o are broken down into their individual components- oxygen, carbon, and hydrogen- and then rearranged to create glucose using energy from the sun. In the Calvin Cycle, the goal is to produce G3P, from CO2, which will eventually become glucose, or sugar, however, this can’t be done without NADPH.
Research discovered by Professor Yutaka Amao, stated that CO2 could be reduced by mimicking this process and can reduce CO2 by combining it with organic compounds. While fumaric acid is typically synthesized from petroleum to be used as a raw material for making biodegradable plastic, this research team was successful in synthesizing fumaric acid, from CO2, powered by sunlight. This process is known as artificial photosynthesis.
It is really interesting how mimicking the process of photosynthesis can lead to CO2 being reduced when combined with organic compounds, and used as raw materials, which can be converted into sustainable structures such as plastic!



Leave a Reply